With more than 100 million gallons of hydraulic oil released to the North American environment every year, it’s no wonder that acceptance of EAFs continues to broaden among users and within industry standards. Moreover, incentives and regulations provided by governments across the globe help encourage the use of biodegradable hydraulic and pneumatic oils. But who’s using EAFs? What applications do they work well in? What are the trade-offs? Can they protect equipment as well as currently used oils? No responsible manager will change to an environmentally acceptable oil if it means sacrificing equipment reliability.
Early generations of biodegradable hydraulic oils were often blends of lower-quality vegetable oils mixed with additives that were developed for petroleum-based oils. These additives often contained compounds that hindered the oil’s natural biodegradability. The result was a hydraulic fluid with poor performance at low temperatures, a short service life, and environmental characteristics that were only marginally better than those of the less-expensive petroleum product that it was replacing.
Now, thanks to advances in base oils and additives, EAFs are bringing much more to the table. New synthetic oils are highly biodegradable and can be used at temperature extremes. Selective breeding and genetic modification have resulted in vegetable-seed oils with the same service life as mineral oils. The development of new antiwear compounds has allowed additive manufacturers to develop products with extremely low toxicity and high biodegradability. These new, nontoxic additives have the same performance as older additives that contain zinc.
In 1999, the U. S. Corps of Engineers coined the term “Environmentally Acceptable Fluids” as those that were readily biodegradable and nontoxic to marine organisms. Since then, this term has been adopted by international standards organizations and the U. S. Environmental Protection Administration, and the requirement that the product not be bioaccumulative was added.
What is environmentally aware?
First of all, let’s look at the criteria for EAF: biodegradation, toxicity, and bioaccumulation.
Biodegradation refers to the ability of bacteria in soil and water to use the lubricant as a food source. The oil is broken down into units small enough to become a source of carbon for bacteria. In the popular ASTM D5864 test, the oil sample is mixed with water and bacteria and incubated for 28 days. Once completed, the sample is evaluated to see what portion was degraded by the bacteria, and what portion remains intact. Today’s enhanced base oils can achieve biodegradation rates of greater than 90% after the 28-day period.
Biodegradation measurements break into two standard criteria. Readily biodegradable are oils that biodegrade by 60% after 28 days. Inherently biodegradable products are those that biodegrade by at least 20%. Both synthetic esters and vegetable oils are considered “readily biodegradable” under this criterion.
Toxicity refers to the lethality of a given concentration of lubricant to a specified organism. Hydraulic or pneumatic-oil toxicity tests usually occur in water, using different types of shrimp, minnows, or trout as the subjects.
Bioaccumulation represents the level of concentration for a given material in an organism’s body after repeated exposure. Certain pesticides, for example, become concentrated in animals as they age. Chlorinated products are commonly bioaccumulative.
Types of environmentally acceptable fluids
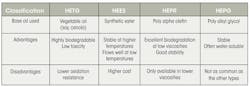
The International Standards Organization (ISO) developed classifications of environmentally acceptable fluids, according to the type of base oil, Table 1. The synthetic ester (HEES) and vegetable oil (HETG) have proven to be the most popular, by a wide margin.
Synthetic esters have been manufactured since the late 1950s. They are made in a chemical reaction between alcohols and acids. Many types of synthetic esters exist, with an array of different characteristics. They feature excellent oxidation resistance and a low pour point, so they can be used over an extended range of application temperatures. In fact, some synthetic esters offer such high stability at high temperatures that they’re used as lubricants in jet engines. Still, due to their molecular structure, microbes in air and water can break the oils down under the right conditions. Synthetic esters, in general, provide good environmental characteristics.
Vegetable seed oils, or natural esters, are pressed from seeds such as soy or rape (commonly known as canola). Naturally occurring, highly biodegradable, and of low toxicity, vegetable oils are ideal for use as lubricant base oils in many applications. Traditionally, though, the use of vegetable oils has been limited to shorter service life and lower temperatures, due to the lower oxidation stability of vegetable oils. Advances in plant genetics have yielded more stable and better oils in recent years. The newest natural ester oils have service lives that equal those obtained with mineral oil fluids.
Beyond their environmental characteristics, vegetable-seed oils feature excellent lubricity and very-high viscosity index. They also typically maintain exceptionally high fire points. Performance does suffer at very low or very-high temperatures, though, because of poor oxidation stability and a relatively high pour point.
EAFs and hydraulic industry standards
Today’s environmentally aware hydraulic fluids, both vegetable oil and synthetic products, can easily meet or exceed most industry standards, such as:
• Vickers M-2950-S,
• Vickers 1-286-5,
• Vickers 35VQ-25 and V-104C (ASTM D-2882) vane pump stand tests,
• Vickers M-2950-S, I-286-S,
• U. S. Steel 126 and U.S. Steel 127,
• Cincinnati Milacron P-68, P-70,
• DIN 51524 Part 2 load stage 10, and
• Denison HF-0, HF-1, HF-2.
Environmentally acceptable hydraulic fluids are typically used in situations where an oil spill would be small, but difficult to prevent, such as older mobile hydraulic systems. Other instances would be where a spill may be rare, but would pose a serious environmental or public relations problem (e.g., an offshore oil platform or hydraulic systems on equipment operating in urban or residential areas).
Primary markets for hydraulic EAFs include forestry, mining, marine and offshore, hydroelectric dams, and mobile hydraulic systems that operate in urban or residential areas. Synthetic esters should be considered in applications that run at high or low temperatures, for OEM original fill, or for hydraulic applications that require longer service life. Such applications take advantage of the synthetic ester’s unique characteristics, thus justifying their higher cost.
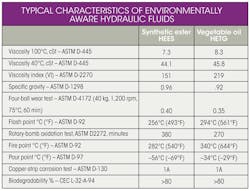
Comparing the fluids
The characteristics of the HETG natural ester often surpass those of the HEES synthetic ester (Table 2). At high temperatures, though, the synthetic fluid offers far better oxidation stability and, therefore, service life. And at very-low temperatures, the synthetic ester flows freely, while the vegetable oil product solidifies at about −30°F.
Hydraulic fluid spills come in various forms. They can occur over time, with a drop-by-drop loss of a quart of oil per week. On the other hand, it might be an immediate, rapid spill, such as a hydraulic hose bursting on an oil platform or the release of oil on heavy equipment that continues until a pump can be turned off.
In the U. S., spills of EAFs must be reported to the National Response Center, U. S. Coast Guard, and local authorities — just as for spills of mineral oils. Generally, EAFs are less toxic and faster to biodegrade than spills of mineral oil lubricants. Cleanup and remediation requirements and costs are normally lower for spills of EAFs than for spills involving mineral oils.
Environmental laws have driven these changes, and in some cases push new, more-restrictive laws. For example, North Sea drilling platforms require that a hydraulic fluid must meet certain environmental standards, which vary depending on the quantity used and the location. Every passing year seemingly brings more hydraulic and pneumatic oil applications under tighter environmental restrictions.
Conclusion
Regulations propel the development and application of enhanced environmentally acceptable fluids. Thanks to new base oils and additives, the latest-generation products are able to meet performance standards that were once only relegated to mineral oils. Equipment owners now can protect both their hydraulic equipment as well as the environment by choosing the right product for their application.
David Sundin, Ph.D., is a chemist, engineer, and founder of SVB Environmental Lubricants, Tyler, Tex. He can be reached at (903) 231-3141 or visit his Web site at www.davidsundin.net.